Synovo Hip Implants: FDA Alert About Risks
Urgent Alert: FDA Warnings on Synovo Hip Implants
The U.S. Food and Drug Administration (FDA) has issued a critical warning in January of 2024 against the use of Synovo Total Hip Replacement Implants due to significant unauthorized modifications. These changes have raised serious concerns about the safety and efficacy of these medical devices, originally cleared for use in 1991. If you or someone you know has undergone hip replacement surgery involving Synovo implants, it’s crucial to be aware of the latest FDA findings and understand the potential risks involved.
What Patients Need to Know
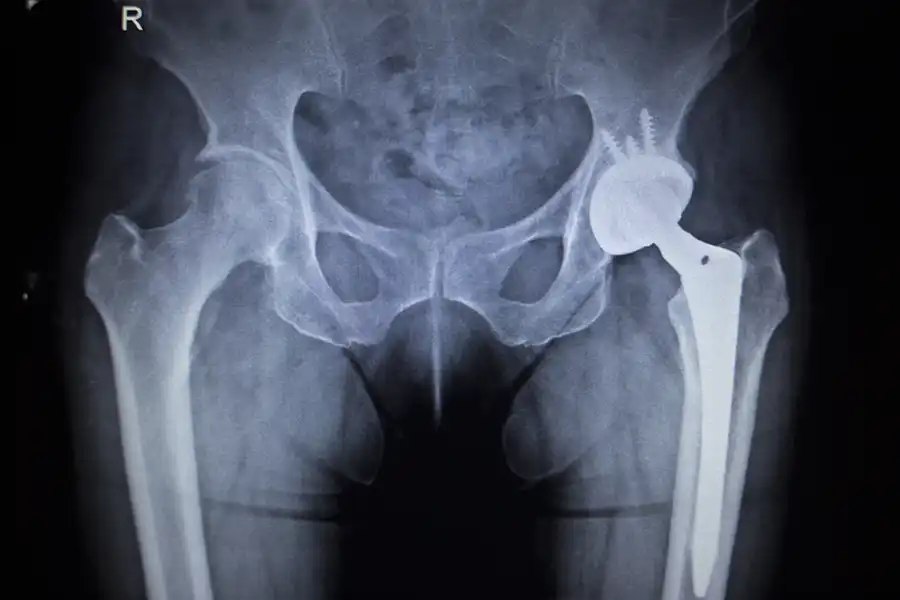
The FDA’s investigation revealed that the Synovo Total Hip System, including key components like the femoral resurfacing cup, acetabular fixation cup, and acetabular bearings, has been significantly modified without approval. These modifications may compromise the implant’s stability and longevity, increasing the risk of implant loosening, pain, and the potential need for revision surgery. The FDA strongly advises healthcare providers to cease the use of these implants and recommends patients with the Synovo system to closely monitor for any new or worsening symptoms.
Potential Risks and Injuries
Patients with the Synovo Total Hip System may face several health risks due to the unauthorized modifications. The primary concerns include increased chances of implant loosening, which could lead to pain, proximal femur fractures, or component fractures. Such complications may necessitate additional, often more complex, revision surgeries. If you experience new or worsening pain, grinding noises, or difficulty bearing weight on the affected side, these could be signs of implant failure and should be addressed immediately with a healthcare provider.
Contact Alonso Krangle, LLP for Legal Support
If you’ve experienced complications or injuries due to a Synovo Total Hip Replacement Implant, Alonso Krangle, LLP is here to help. Our experienced legal team specializes in medical device litigation and is committed to advocating for the rights of patients affected by faulty implants. Contact us today at [PHONE] for a free consultation to discuss your case and explore your legal options. Don’t wait to seek the justice and compensation you deserve; let Alonso Krangle, LLP guide you through every step of the legal process. Call us today at [PHONE].
Speak with An Attorney
Submit This Form or Call 800-403-6191

