Posts by attorney
Is It Too Late to File a Cartiva Lawsuit? Legal Deadlines Explained
The Deadline to File a Cartiva Lawsuit Many people injured by the failed Cartiva Synthetic Cartilage Implant (SCI) are only now learning about the device’s 2024 recall and the growing number of lawsuits filed against Stryker, the manufacturer. But when it comes to taking legal action, time is critical. Every state limits how long injured…
Read MoreCartiva vs Toe Fusion – Why Surgeons Now Prefer Fusion After Cartiva Failures
Cartiva vs Toe Fusion – Why Surgeons Are Returning to a Proven Approach For years, the Cartiva Synthetic Cartilage Implant was promoted as a breakthrough in treating big toe arthritis, or hallux rigidus. It promised motion preservation, faster recovery, and lasting pain relief-something traditional toe fusion could not offer. But after thousands of failures and…
Read MoreThe Hidden Costs of a Failed Cartiva Implant – Pain, Lost Work, and Endless Medical Bills
The Real Cost of a Failed Cartiva Implant The pain of a failed Cartiva Synthetic Cartilage Implant goes far beyond the joint itself. When the device fails, patients face a ripple effect that reaches every corner of their lives-mounting medical bills, time away from work, and ongoing physical and emotional stress. What began as a…
Read MoreInside the Cartiva Lawsuits – What Lawyers Are Discovering About Device Failures
What the Cartiva Lawsuits Are Revealing About Device Failures The wave of lawsuits filed over the Cartiva Synthetic Cartilage Implant is doing more than seeking compensation for injured patients-it’s exposing what really went wrong behind the scenes. As law firms dig into company documents, surgeon reports, and regulatory filings, a troubling picture is emerging. Thousands…
Read MoreHow to Tell if Your Cartiva Implant Is Failing (and What to Do About It)
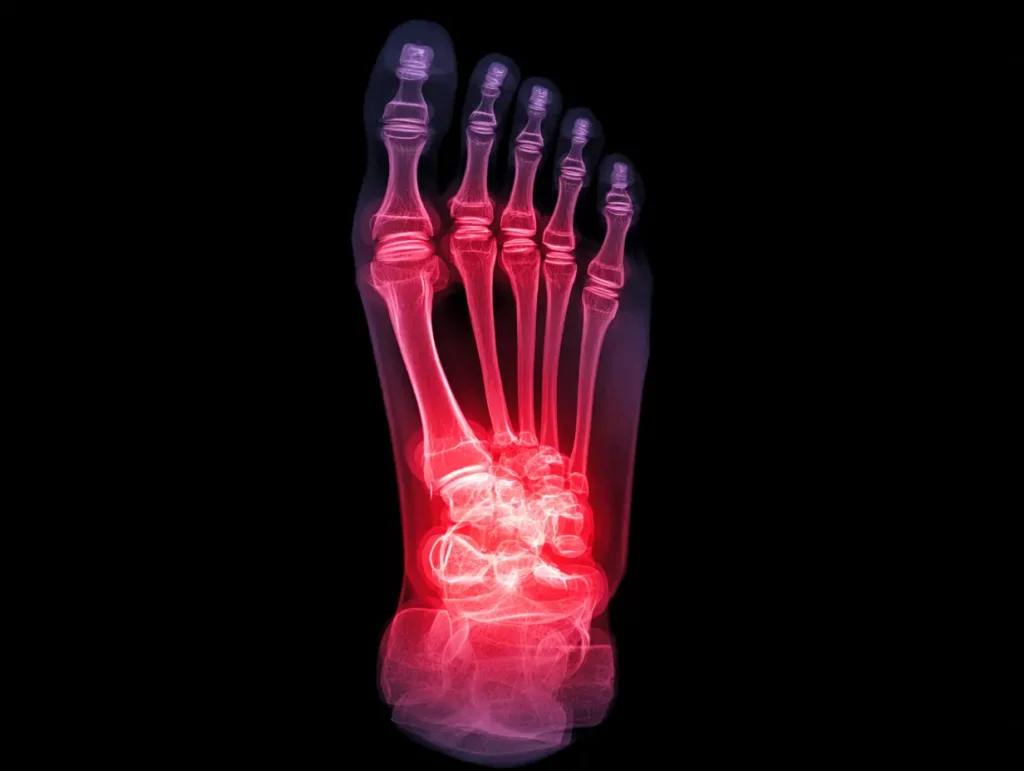
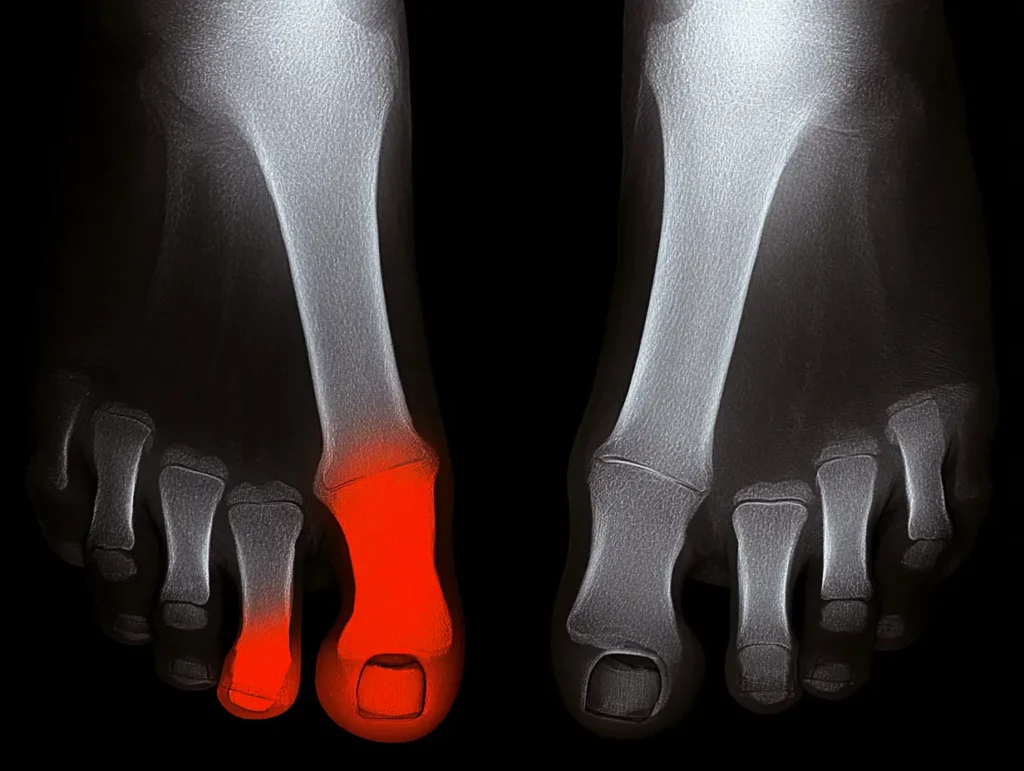
Recognizing the Signs of a Failing Cartiva Implant When the Cartiva Synthetic Cartilage Implant was first introduced, it was marketed as a durable, motion-preserving alternative to toe fusion for arthritis in the big toe joint. Unfortunately, many patients are now experiencing painful complications months or even years after surgery. If you received a Cartiva implant,…
Read MoreFrom Innovation to Injury – The Rise and Fall of the Cartiva Toe Implant
The Rise and Fall of the Cartiva Toe Implant When the Cartiva Synthetic Cartilage Implant was first introduced, it was hailed as a major step forward in orthopedic medicine-a simple, less invasive way to restore mobility for patients suffering from arthritis in the big toe joint. It promised the best of both worlds: relief without…
Read MoreWhy the Cartiva Recall Could Be Bigger Than Anyone Expected
The Expanding Fallout from the Cartiva Recall When Stryker Corporation announced a nationwide recall of the Cartiva Synthetic Cartilage Implant in late 2024, it seemed at first like a limited correction-a product quietly withdrawn due to “performance concerns.” But as more information emerges, it’s becoming clear that this is no ordinary recall. The number of…
Read MoreEmerging Mass Tort: Dupixent and the Link to Rare Skin Cancer (CTCL)
Emerging Mass Tort: Dupixent and the Link to Rare Skin Cancer (CTCL) The FDA’s safety investigation into Dupixent and its potential link to Cutaneous T-Cell Lymphoma (CTCL) has created a storm of concern among patients, physicians, and attorneys nationwide. Dupixent (dupilumab) has long been marketed as a breakthrough for moderate to severe atopic dermatitis. But…
Read MoreDupixent and Cancer: Understanding the Four-Fold Increased Risk
Dupixent and Cancer: Understanding the Four-Fold Increased Risk Recent medical findings have raised alarming questions about the safety of Dupixent, a biologic medication prescribed for chronic skin and respiratory conditions. Data emerging from post-market studies and adverse event reports suggest that patients taking Dupixent may face a four-fold increased risk of developing a rare type…
Read MoreHave You Suffered a Dupixent Injury? Recognizing the Warning Signs of CTCL
Have You Suffered a Dupixent Injury? Recognizing the Warning Signs of CTCL Dupixent injury cases are becoming more common as patients report worsening skin conditions months or even years after beginning treatment. For many, Dupixent initially brings tremendous relief from eczema or dermatitis, but over time, some begin to experience persistent, painful, or changing rashes…
Read More