Medical Device
Synovo Hip Implants: FDA Alert About Risks
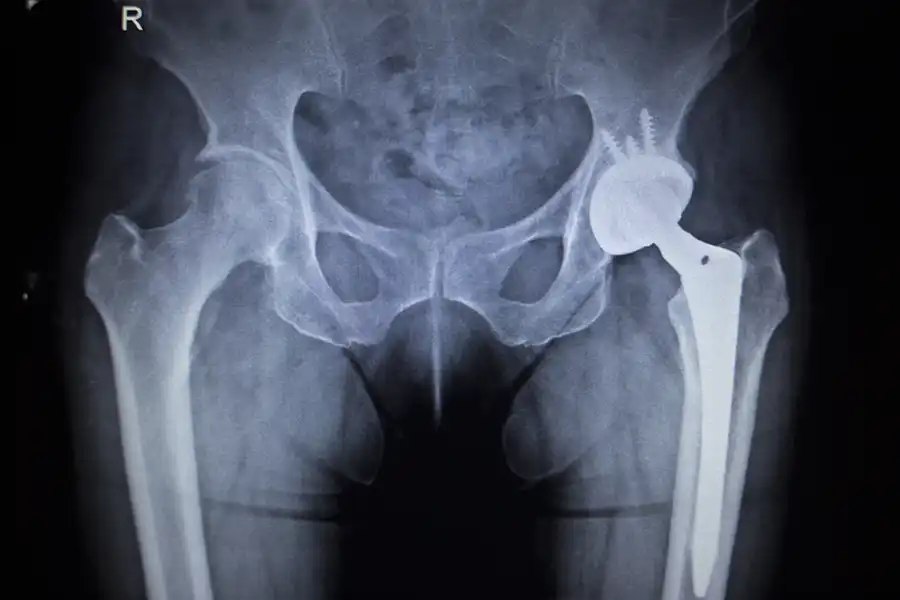
Urgent Alert: FDA Warnings on Synovo Hip Implants The U.S. Food and Drug Administration (FDA) has issued a critical warning in January of 2024 against the use of Synovo Total Hip Replacement Implants due to significant unauthorized modifications. These changes have raised serious concerns about the safety and efficacy of these medical devices, originally cleared…
Read MoreFDA Warning on Synovo Hip Implants
Understanding the FDA Warning: The Risks of Synovo Total Hip Replacement Implants The U.S. Food and Drug Administration (FDA) has recently issued a significant warning to healthcare providers and patients regarding the use of Synovo Total Hip Replacement Implants. This advisory comes in light of discoveries that substantial modifications have been made to the system’s…
Read MoreThe Unseen Dangers of Bard PowerPort: A Comprehensive Guide to Legal Recourse
Understanding the Bard PowerPort The Bard PowerPort, a product of medical equipment manufacturer Bard, is an implanted port device, also known as a port catheter or port-a-cath. This device, typically inserted under the skin of the chest, consists of a small reservoir, or port, connected to a thin catheter. It is designed to simplify the…
Read MoreUnderstanding Bard PowerPort Lawsuits: A Comprehensive Guide
The Bard PowerPort Catheter The Bard PowerPort is a medical device, often referred to as a port catheter or port-a-cath, that is implanted under the skin of the chest. This device, connected to a thin catheter, is designed to simplify the administration of injectable fluids, blood transfusions, chemotherapy, and other medications. Despite its intended benefits,…
Read MoreUnderstanding the Bard PowerPort Lawsuits: A Comprehensive Guide
Introduction to Bard PowerPort Catheters A port catheter, often referred to as a port-a-cath, is a device implanted under the skin, typically in the chest area. This device consists of a small reservoir, known as a port, connected to a thin catheter. It simplifies the administration of injectable fluids, blood transfusions, chemotherapy, and other medications.…
Read MoreUnraveling the Bard PowerPort Lawsuit: An In-Depth Examination
Unraveling the Intricacies of the Bard PowerPort Lawsuit: An In-Depth Examination In the realm of medical devices, the Bard PowerPort has emerged as a significant point of contention. This implantable port catheter device, designed for the streamlined administration of intravenous fluids or medication, has been the subject of numerous product liability lawsuits. This comprehensive guide…
Read MoreUnderstanding the Bard PowerPort Lawsuits: A Comprehensive Guide
Understanding the Bard PowerPort: A Comprehensive Guide The Bard PowerPort is a medical device that has been the subject of numerous discussions and legal actions due to its alleged defects and the complications it may cause. This article aims to provide a detailed understanding of the PowerPort, its uses, potential risks, and the complications that…
Read MoreUnderstanding the Bard PowerPort Device and Its Complications
Understanding the Bard PowerPort Device and Its Complications The Bard PowerPort, a product of Bard Access Systems, Inc., is an implantable port catheter device designed to provide convenient access to a patient’s veins for the administration of medication and fluids. While this device has been instrumental in streamlining medical procedures, it has also been associated…
Read MoreBard PowerPort Lawsuits: Understanding Device Failures, Injuries, and Legal Actions
Introduction to Bard PowerPort The Bard PowerPort, a product of Bard Access Systems, Inc., is an implantable port catheter device designed to provide convenient access to a patient’s veins for the administration of medication and fluids. This device, approved by the FDA in 2000, was initially hailed as a game-changer in the realm of medical…
Read MoreCovidien Mesh Lawsuits and Potential Multi-District Litigation
Covidien Mesh Lawsuits and Potential Multi-District Litigation With thousands of claims in state court and 73 cases filed in federal courts pertaining to its hernia mesh device, Covidien again seeks to consolidate claims into a multi-district litigation (MDL). In June of 2020, the manufacturer’s request for the creation of an MDL was denied, as there…
Read More