Paragard Bellwether Trial
The Paragard Bellwether Trial
The Paragard bellwether trial represents a significant turning point for individuals claiming injuries due to the Paragard IUD. Bellwether trials play a critical role in large multidistrict litigation (MDL) cases, helping to test legal arguments and set benchmarks for settlement negotiations in similar cases. For the thousands of plaintiffs alleging injuries from the Paragard intrauterine device (IUD), the bellwether trial will help establish whether their cases can be successfully pursued and what types of damages may be recoverable.
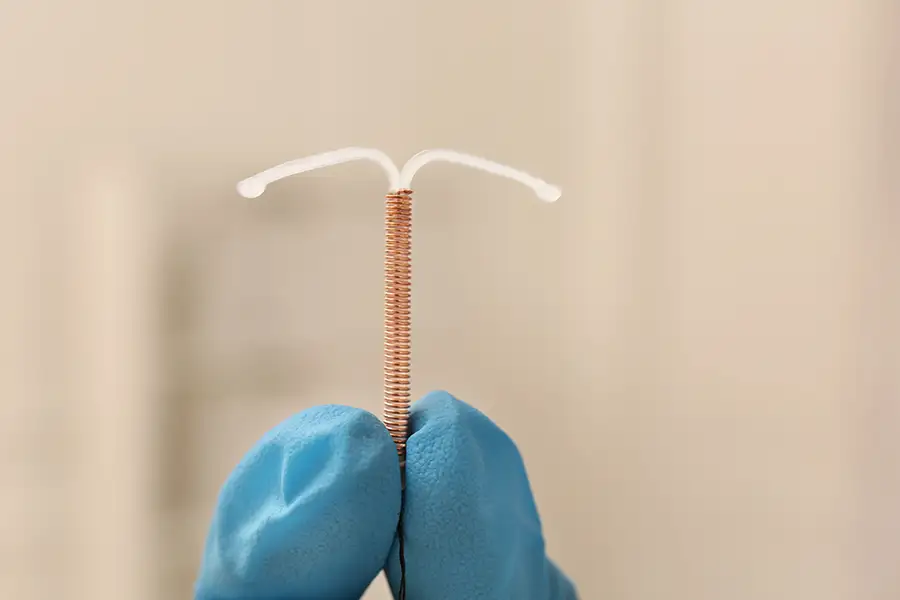
The upcoming bellwether trial will specifically address allegations that the Paragard IUD, manufactured by Teva Pharmaceuticals, poses serious risks to users due to its design and lack of adequate warnings. As of December 2025, the first Paragard bellwether trial will allow attorneys to present evidence of the injuries and complications reported by claimants. This trial is crucial, as it sets a potential pathway for other cases within the MDL to reach resolution more efficiently.
Understanding the stakes and procedural steps in the Paragard bellwether trial is essential for plaintiffs and observers alike. Here, we discuss what to expect from this trial and its potential impact on the legal landscape surrounding medical device liability.
Background on the Paragard IUD and Reported Issues
The Paragard IUD is a non-hormonal contraceptive device made of copper that has been available for decades as an alternative to hormone-based IUDs. Unlike hormonal IUDs, Paragard relies on copper to prevent pregnancy. Despite its popularity, Paragard has faced increasing scrutiny as reports of adverse effects have mounted. Plaintiffs in the Paragard lawsuits claim that the device is prone to breaking during removal, causing severe internal injuries, including perforations, infections, and, in some cases, lasting reproductive damage.
The basis for the lawsuits involves allegations that Teva Pharmaceuticals failed to adequately warn patients of these risks. Many claimants allege that they were not informed of the device’s potential for breakage or the complications that could arise during removal. This has led to an influx of lawsuits consolidated into the MDL, which aims to handle the cases more efficiently by addressing common legal and factual issues in a single court.
The stakes of the Paragard bellwether trial are high, as the case will assess whether Teva could have reasonably anticipated these risks and if the product’s design is defective. For thousands of affected individuals, this trial is an essential step toward accountability and potential compensation for their injuries.
Development of Paragard MDL and Purpose of Bellwether Trials
The Paragard litigation has been consolidated into MDL No. 2974 in the U.S. District Court for the Northern District of Georgia, presided over by Judge Leigh Martin May. MDLs like this one streamline the legal process by combining similar cases involving complex issues, such as medical device defects. This allows the court to efficiently address large numbers of claims while offering consistency in rulings on evidence and procedural issues. Judge May’s role includes managing the litigation, setting key dates, and overseeing pretrial motions.
The purpose of a bellwether trial is to serve as a test case that provides insights into the strengths and weaknesses of the claims, as well as the defenses presented by Teva Pharmaceuticals. If the plaintiffs are successful, this outcome could pave the way for favorable settlements or verdicts in similar cases. Conversely, a ruling in favor of the manufacturer may influence settlement discussions and the trajectory of the MDL as a whole.
With the Paragard bellwether trial scheduled for December 2025, this test case will allow all parties to evaluate the viability of claims in front of a jury, potentially guiding the outcome for thousands of Paragard-related lawsuits.
Timeline and Key Milestones Leading Up to the Bellwether Trial
The Paragard bellwether trial is set to commence in December 2025, following a structured timeline that has included crucial preparatory steps for both plaintiffs and defendants. Since the MDL’s formation, important milestones have been established, starting with the initial case filings and progressing through discovery phases, depositions, and expert witness selections.
Key dates have included deadlines for evidence collection, pretrial motions, and decisions on admissible expert testimony. Judge May has also set rulings on procedural matters that shape how evidence will be presented in court, a process critical to ensuring fairness for both parties. This groundwork has involved intensive document review, interviews, and expert analysis, all aimed at preparing a strong foundation for the bellwether trial.
The approach to pretrial activities underscores the importance of thorough preparation in complex litigation like the Paragard cases. By establishing clear guidelines and deadlines, the court has enabled both sides to streamline their cases and avoid unnecessary delays. This organized approach not only serves to expedite the trial process but also encourages informed settlement discussions that may follow the trial.
Primary Legal Issues in Paragard Litigation
The Paragard bellwether trial will focus on several core legal issues, with plaintiffs primarily arguing that the Paragard IUD is defective in design and that Teva Pharmaceuticals failed to provide adequate warnings. These allegations bring up critical points under product liability law, where manufacturers are held responsible for ensuring the safety of their products and adequately informing consumers of any risks.
One of the central arguments is that the Paragard device’s design may contribute to its tendency to break during removal, leading to severe complications for users. Plaintiffs argue that this defect could have been identified and addressed by the manufacturer before reaching the market. Furthermore, there is an assertion that the warning labels and instructions provided with the device did not adequately inform users and medical professionals of the risks associated with device removal.
Defendants, on the other hand, may contend that Paragard’s approval by the U.S. Food and Drug Administration (FDA) supports its safety profile and that any breakage incidents stem from improper handling or rare, unpredictable complications. This line of defense highlights the challenges inherent in proving that a product defect, rather than external factors, caused the alleged injuries.
Injuries and Damages Claimed by Paragard Plaintiffs
Individuals involved in the Paragard lawsuits report a range of injuries primarily associated with device breakage during removal. Common injuries include uterine perforation, internal infections, pelvic pain, and in severe cases, permanent reproductive harm. These complications often require surgical interventions to retrieve device fragments, adding to the physical and emotional toll on patients.
In the bellwether trial, plaintiffs seek compensation for these injuries under several categories of damages. Economic damages encompass medical expenses, including costs for surgery, follow-up care, and any long-term treatment resulting from complications. Additionally, plaintiffs may claim compensation for lost wages if their injuries impeded their ability to work.
Non-economic damages, including pain and suffering and emotional distress, are also anticipated. Some plaintiffs may seek punitive damages, intended to hold Teva accountable for allegedly ignoring risks associated with Paragard. This pursuit of damages reflects both a desire for personal recovery and a broader call for accountability from medical device manufacturers.
Expert Testimonies and Scientific Evidence in the Trial
Expert testimony is a cornerstone of the Paragard bellwether trial, as both sides rely on medical and scientific experts to support their claims. Plaintiffs will present expert witnesses, including gynecologists and product engineers, to testify on the alleged design flaws and health risks of the Paragard IUD. These experts are expected to discuss how device breakage might occur during removal and the potential health impacts of such incidents.
Scientific studies are also a key component of the trial evidence. For instance, research published in medical journals and studies from the National Center for Biotechnology Information (NCBI) examine IUD safety, device longevity, and removal complications. Plaintiffs may reference these studies to bolster their claims that the Paragard device has a higher risk profile than publicly disclosed.
For the defense, expert witnesses are likely to counter by emphasizing the Paragard’s general safety and FDA approval. They may also present data suggesting that removal complications are rare and argue that breakages may result from specific handling techniques rather than inherent design flaws. This testimony will be crucial in shaping the jury’s understanding of complex medical issues at the heart of the case.
Potential Outcomes of the Bellwether Trial and Implications for Other Cases
The results of the Paragard bellwether trial could have far-reaching consequences for the thousands of cases consolidated under the MDL. A favorable outcome for plaintiffs may encourage Teva Pharmaceuticals to consider settlements, potentially leading to financial compensation for many claimants. Such a result could also signal the strength of plaintiffs’ claims and influence future product liability lawsuits.
Alternatively, if the verdict favors the defense, this could undermine plaintiffs’ bargaining power in settlement discussions and discourage some cases from advancing. However, each trial is unique, and a single outcome does not necessarily determine the result of other cases within the MDL. Bellwether trials provide insights, but each plaintiff retains the right to pursue individual claims or participate in broader settlement agreements.
In either scenario, the bellwether trial will likely serve as a precedent, guiding future Paragard litigation and setting expectations for compensation or legal strategies in similar product liability cases.
Comparison to Other IUD-Related Product Liability Cases
The Paragard lawsuits are not the first involving allegations of defective IUDs. Other well-known cases, such as those involving the Mirena IUD, have set notable precedents in product liability and medical device safety. In the Mirena litigation, plaintiffs also claimed injuries from device migration, perforation, and inadequate warning labels, similar to the issues cited in the Paragard cases.
Analyzing the outcomes of past IUD-related cases provides valuable insight into potential paths for the Paragard litigation. For instance, many IUD lawsuits have resulted in settlements, allowing plaintiffs to recover damages without prolonged court battles. However, differences in device design, medical technology, and specific injury profiles mean that each case must be evaluated independently.
The similarities and differences between Paragard and other IUD cases highlight the challenges inherent in holding medical device manufacturers accountable while navigating complex legal and scientific landscapes.
What Plaintiffs Should Know as the Trial Approaches
As the Paragard bellwether trial date nears, plaintiffs involved in the MDL should stay informed of updates that could impact their cases. Key developments, including pretrial motions, expert testimonies, and procedural rulings, may influence the direction and outcome of the MDL. Regular updates from legal counsel are critical to ensure that plaintiffs understand their rights and the likely trajectory of their cases.
For plaintiffs awaiting trial, knowing what to expect can help ease the uncertainty often associated with prolonged litigation. Working closely with experienced product liability attorneys can be instrumental in navigating these complex proceedings. Legal representatives can advise on case status, potential settlements, and other relevant developments.
Additionally, plaintiffs should understand that even if the bellwether trial does not end in a settlement, it could still provide valuable insights that strengthen their cases. As the trial approaches, claimants should feel reassured by the legal process and the support of their legal teams dedicated to seeking accountability on their behalf.
Role of the Judge and the Court in Shaping Paragard Outcomes
The presiding judge in the Paragard MDL, Judge Leigh Martin May, plays an essential role in guiding the litigation process. Judge May has overseen all procedural aspects of MDL No. 2974, including case consolidation, evidence admissibility, and scheduling, ensuring that the trial is fair and balanced for both plaintiffs and the defendant. Her rulings on pretrial motions and evidence admissibility are especially significant, as these decisions shape what information the jury will consider.
Judge May’s oversight of expert witness qualifications and scientific evidence admissibility ensures that only reliable information reaches the jury. In high-stakes product liability trials, the court’s role extends beyond maintaining order to influencing how evidence is presented and interpreted. Her rulings could set a standard for future MDL trials and similar lawsuits involving medical devices.
Through her rulings and case management, Judge May provides a framework that allows the bellwether trial to move forward efficiently, giving both parties a fair opportunity to present their cases. Her role in maintaining procedural consistency across multiple cases within the MDL is critical for ensuring justice in a complex, large-scale litigation.
The Significance of the Paragard Bellwether Trial for Public Health and Safety
Beyond the individual plaintiffs’ cases, the Paragard bellwether trial carries broader implications for public health, patient safety, and the responsibilities of medical device manufacturers. The trial highlights the importance of transparent communication about medical device risks, emphasizing that patients have a right to be informed about potential complications before undergoing procedures.
If the court finds that Teva Pharmaceuticals failed to adequately warn users about the risks associated with Paragard, it could prompt regulatory reviews or stricter guidelines for device approval and patient disclosure. This trial may serve as a warning to other manufacturers to prioritize product safety and clear communication, potentially leading to improvements in how medical devices are evaluated and marketed.
The trial underscores a critical conversation on medical device accountability and patient rights, illustrating that legal action can influence not only individual compensation but also broader public health practices. For consumers, the Paragard litigation serves as a reminder of the importance of informed medical decisions and the role of legal recourse in addressing medical harm.
What’s Next for Paragard Plaintiffs and the Bellwether Trial
The Paragard bellwether trial marks a pivotal moment in the journey for plaintiffs seeking justice for their alleged injuries. As the December 2025 trial approaches, plaintiffs and their legal teams remain focused on presenting a strong case that demonstrates the risks and injuries tied to the Paragard IUD. The outcome of this trial could shape the direction of thousands of similar cases, potentially leading to settlements or additional trials depending on the verdict.
For claimants, this trial offers an opportunity for their voices to be heard, with the possibility of compensation that acknowledges the physical, emotional, and financial toll of their injuries. Given the significance of the trial, plaintiffs are encouraged to stay informed and proactive, consulting with their legal counsel as updates unfold.
If you or a loved one has experienced complications with the Paragard IUD, contact Alonso Krangle, LLP today. Our attorneys are here to help you understand your options and provide guidance throughout the litigation process. Call 800-403-6191 or fill out the online form to schedule a consultation with our team.
Speak with An Attorney
Submit This Form or Call 800-403-6191

